| Title | Tentech Inc. Receives UAE MoH Certification for 10THERMA | ||
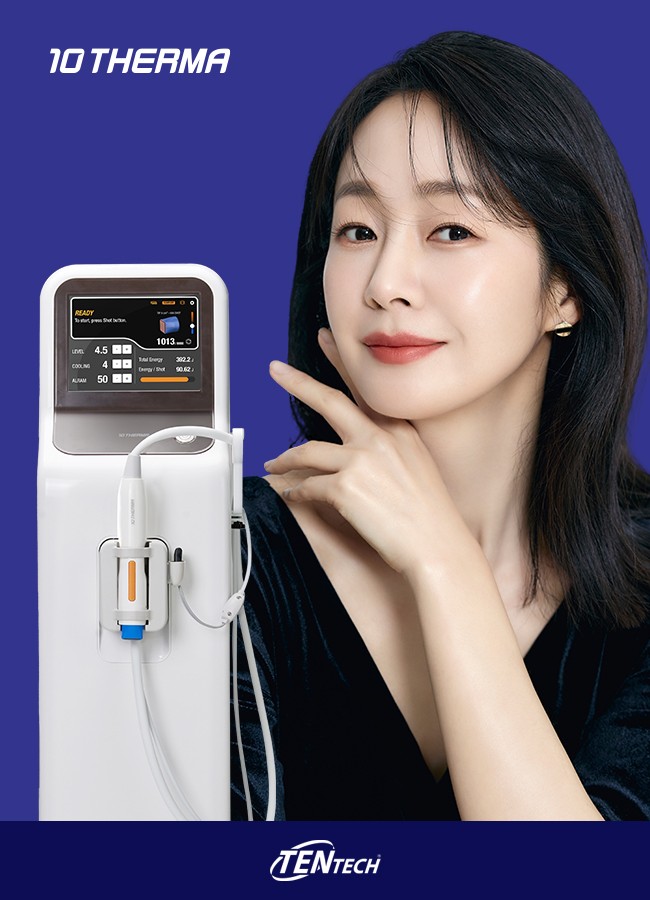
Tentech Inc., a medical device manufacturer, announced on Tuesday that it has received medical device certification for 10THERMA from the UAE. Ministry of Health (MoH). 10THERMA, with a maximum output of 400W, is a 6.78MHz monopolar radiofrequency (MRF) treatment device. 10THERMA can deliver RF energy to the dermis, generating heat through the skin's electrical resistance. The 5㎠ face tip is wider than those of other RF devices, allowing for 20% more energy coverage per shot. 10THERMA features an S.T.C. (Smart Temperature Cooling) system, which emits a total of seven cooling pulses during a single shot. Tentech Inc. has already secured certifications for 10THERMA from several global regulatory agencies, such as the United States (U.S. FDA), Taiwan (TFDA), Vietnam (MoH), Indonesia (MoH), Thailand (TFDA), and Malaysia (MDA). Meanwhile, Tentech Inc. has been making consistent efforts to build brand awareness for 10THERMA and Tentech’s other products by participating in exhibitions and conferences held in the Middle East, such as 'Dubai Derma 2024' and 'Arab Health 2024' in the first quarter of 2024. A representative for Tentech Inc. stated, "10THERMA sales have surpassed 700 units, gaining popularity internationally." He added, "We plan to officially launch 10THERMA in the UAE. by developing a market-specific strategy based on the local market conditions in the UAE." |
|||
| Hits | # 152 | Date | 2024.05.28 |
